Common methods of brain imaging present many challenges: some are invasive, others may contain harmful radiation, and many are difficult to use or require the patient to remain absolutely still throughout the process. Today researchers are developing non-invasive methods such as a type of spectroscopy that uses IR to measure blood flow in the brain, a new light-sheet fluorescence tool that images brain activity in 3D, and a wearable cap with embedded IR lasers that can be worn by infants to detect brain damage.
Near-IR measures blood flow in the brain
Measuring cerebral blood flow is important for diagnosing strokes, and for predicting secondary damage in subarachnoid hemorrhages or traumatic brain injuries. Doctors who provide neurological intensive care, would also like to monitor a patient’s recovery by imaging brain blood flow and oxygenation.
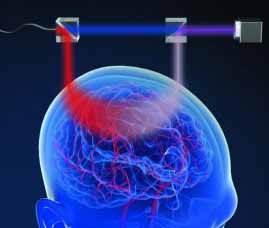
Functional interferometric diffusing wave spectroscopy is a noninvasive method currently under development that uses near-infrared light to measure blood flow in the brain. This is a less costly method that can be used for assessing brain injuries, compared to MRI and CT scanners, and it is being developed by biomedical engineers and neurologist at the University of California, Davis.
The researchers found that they could use the new technology to measure blood flow more rapidly and deeper below the surface than with current light-based technology (see photo above). They could measure pulsating cerebral blood flow and could also detect changes when volunteers were given a mild increase in carbon dioxide.
“Now we can assess how well the brain regulates blood flow, and even detect brain activation noninvasively in adult humans, using principles similar to functional magnetic resonance imaging (fMRI), but at a fraction of the cost,” said Vivek Srinivasan, adjunct associate professor of biomedical engineering at UC Davis and senior author on the study.
For more, see Measuring Brain Blood Flow and Activity With Light
Light-sheet fluorescence tool for imaging brain activity
Light-sheet fluorescence imaging shows promise for imaging brain activity in 3D with high speed and contrast. In this technique, a thin sheet of laser light (the light-sheet) is directed through a brain tissue region of interest, and fluorescent activity reporters within the brain tissues respond by emitting fluorescence signals that microscopes can detect. Scanning a light sheet in the tissue enables high-speed, high-contrast, volumetric imaging of the brain activity. Currently, using light-sheet fluorescence brain imaging with nontransparent organisms, like a mouse, is difficult because of the size of the necessary apparatus. To make experiments with nontransparent animals and, in the future, freely moving animals feasible, researchers will first need to miniaturize many of the components.
Researchers from the California Institute of Technology, University of Toronto, University Health Network, the Max Planck Institute of Microstructure Physics, and Advanced Micro Foundry developed a miniature light-sheet generator, or a photonic neural probe, that can be implanted into a living animal's brain. When tested in brain tissues from mice that were genetically engineered to express fluorescent proteins in their brains, the probes permitted the researchers to image areas as large as 240 μm × 490 μm. Moreover, the level of image contrast was superior to that with an alternative imaging method called epifluorescence microscopy.

Describing the significance of his team's work, the study's lead author, Wesley Sacher, says, "This new implantable photonic neural probe technology for generating light sheets within the brain circumvents many of the constraints that have limited the use of light-sheet fluorescence imaging in experimental neuroscience. We predict that this technology will lead to new variants of light-sheet microscopy for deep brain imaging and behavior experiments with freely moving animals."
For more, see Tiny Implantable Tool for Light-Sheet Imaging of Brain Activity
Infrared scanner detects brain damage in infants
No medical tools currently exist to create a harmless, real-time, continuously moving image inside newborn babies' delicate brains. MRI scans can provide an accurate picture inside adults but have drawbacks for use with infants including the requirement for the patient to stay still throughout the process.

What may be a major step forward is a new wearable device developed by the TinyBrains health consortium in conjunction with The Institute of Photonic Sciences in Barcelona. The team put infrared lasers into a tiny cloth cap that can be worn by an infant. Combined with ultrasound pulses, the scientists send harmless signals into the brain of a child. It works almost like an ultrasound scan, but using light to give much more information, a more detailed picture and a much greater resolution.
The signals can measure the cause of many neurodevelopment disabilities by keeping a close eye on any slight drops in critical oxygen levels to and from the brain instantaneously in real-time.
TinyBrains project coordinator, Professor Turgut Durduran, said, “We are using high-density near-infrared spectroscopy (fNIRS) and diffuse correlation spectroscopy (DCS) to measure the oxygen saturation levels in the blood. By integrating both of them with an imaging device as the electroencephalography (EEG), the resulting 3D images have higher resolution, increase the brain specificity and penetration and for the first time, a spatial resolution to this class of measurements.”
By identifying brain function alterations during surgery and stays in intensive care units will allow doctors to analyze why brain disorders frequently occur in the postnatal period and to pinpoint the types of clinical interventions that can improve the neurological outcome of these infants and, ultimately, their quality of life, as infants, young persons, and adults.
For more see, Tiny Brains Project: IR scanner spots brain damage in infants
Written by Anne Fischer, Editorial Director, Novus Light Technologies Today


















 Back to Features
Back to Features