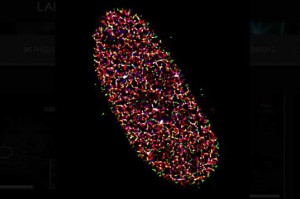
The lab of Professor Eli Rothenberg at the New York University School of Medicine studies the mechanisms of biochemical factors that participate in repair of DNA damage (which may otherwise lead to cancer), and develops new imaging methods that will enable them to visualize the dynamic behavior of individual biological molecules at the nanometer scale.
Prof. Rothenberg and team use super-resolution imaging methods for their research, especially Stochastic Optical Resolution Microscopy (STORM). STORM was first published in 2006 and makes use of photo-switchable fluorophores, rather than traditional photo-activated. These photo-switchable dyes randomly (stochastically) switch from an emitting state (on) to a dark/bleached state (off). This random fluorophore blinking means that by taking many images, all fluorophores can be localized, even if they are closer together than the diffraction limit (~200 nm). This allows for a super-resolution image to be produced after processing many images. STORM is similar to PALM and DNA-PAINT, all are known as super-resolution single-molecule localization microscopy (SMLM) techniques, where fluorophores are temporally separated.
As the fluorophore blinking in STORM is random, each frame of an acquisition a different subset of dye molecules will be emitting. Many of the emitting dye molecules will be far enough apart that they can be individually localized, requiring the processing of thousands of frames into a reconstructed super-resolution image. This means that STORM is highly reliant on the fluorophore properties, from the emitted fluorophore signal intensity to the blinking frequency and density. If the dyes blink too fast, there could be crossover between neighboring dye molecules, preventing individual localization, but if they blink too slow it may not be possible to acquire enough frames to localize every molecule.
Super-resolution imaging
The samples studied by Prof. Rothenberg and team are highly dense, as they are imaging the replication of DNA inside the nucleus of a cell. This means that with traditional microscopy methods it is challenging to clearly resolve the dense crowd of extremely small labeled components. To determine how each component is organized spatially, the lab often uses STORM with three or four colors sequentially, which makes resolution, sensitivity, and localization accuracy a great concern. As outlined by a postdoc in the lab, Dr. Yandong Yin, “We try to look at the nucleus of cancer cells as they are replicating their DNA. The DNA and the proteins involved in DNA replication are labelled so we can understand what is going on when replication happens, but DNA replication fork is very small. We can’t image it without super resolution.”
The need for super-resolution STORM imaging means the choice of detector technology is vital in order to maintain image quality when capturing the many images required for super-resolution reconstruction. Methods of increasing throughput, such as capturing images at a higher speed or across a larger area, would also be beneficial due to the processing time needed to produce super-resolution images.
STORM post-processing conditions are based on the variances of each pixel in camera sensor, correcting for any major variations, in order to better fit the point spread function (PSF) of each fluorophore. Because of this, pixel to pixel variations like those seen in pattern noise on front-illuminated CMOS cameras becomes a major problem. New camera technologies were needed to better tackle super-resolution STORM imaging.
Back-illuminated sCMOS
The lab of Prof. Rothenberg previously used EMCCD technologies, these cameras are highly sensitive due to electron multiplication technology, but featured small sensors and slow speeds, both of which limited throughput. In addition, EMCCD cameras often struggle with factors such as excess noise factor and EM gain decay, making them unsuitable as long term solutions. With EMCCD and front-illuminated CMOS camera technologies limiting imaging, Prof. Rothenberg made the move to back-illuminated sCMOS, a technology ushered in with the Prime 95B.
The Prime 95B features a back-thinned sensor and improved sensitivity thanks to a higher quantum efficiency (a near perfect 95% at peak), improving signal collection, and a lack of pattern noise due to no split sensor, which minimizes the effect of noise. The high sensitivity of the Prime 95B allows for detection of low-emission blinking fluorophores, the importance of which is described by Dr. Yin, “For single-molecule localization imaging, the most important thing is the reconstruction process in how we fit each single-molecule point spread function into its centroid coordinate… If you have a higher sensitivity camera you can have a shorter exposure time, so you can track faster kinetics. More sensitivity and shorter exposure times with the Prime 95B allow us to image faster and track kinetics better.”
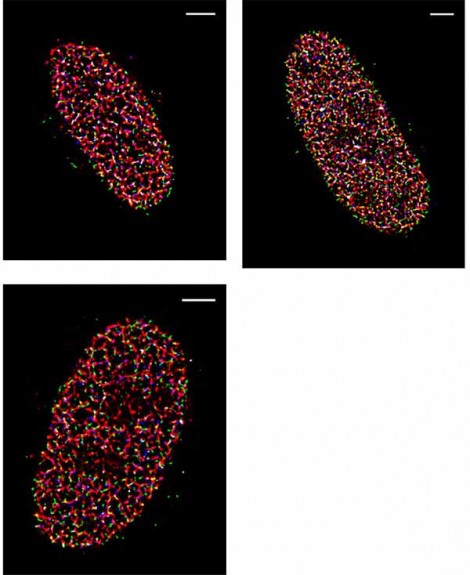
The post-processing of STORM images collected with the Prime 95B are made significantly easier because of the reduction in pattern noise, which makes the localization and reconstruction of fluorophores of multiple colors easier to do. Dr. Yin continued, “The variance for each pixel is much smaller than what is reported on other sCMOS cameras. We have found that more than 90% of the pixels fall within a very tight noise distribution.”
Additionally, the large field of view, high camera speeds, and improved sensitivity allows the team to get a larger image that contains useful information more often. Since Prof. Rothenberg and team often work with samples that are dark at the start of acquisition, it was an issue that they couldn’t guarantee finding their sample and seeing something with the smaller field of view of an EMCCD. Dr. Yin says “Previously we used an EMCCD, but the EMCCD has a smaller chip. With a bigger chip we can see multiple cells simultaneously.”
Written by Dr. Matthew Köse-Dunn, Science Writer, Content Manager and Field Application Specialist, Teledyne Photometrics



















 Back to Enlightening Applications
Back to Enlightening Applications



























