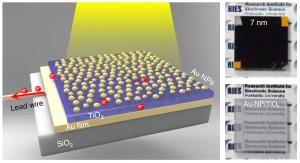
A photoelectrode developed by researchers at Hokkaido University, Japan, can harvest more than a surprising 85% of visible light across a wide spectral range by using a 30-nanometer titanium dioxide (TiO2) thin-film semiconductor sandwiched between a 100-nanometer gold film and loaded with gold nanoparticles to enhance light absorption.
At first, the team had studied the decomposition of water by visible light irradiation with a plasmon-induced water-splitting system based on the localized surface plasmon resonance (LSPR) of gold nanoparticles (Au-NPs) in a Au-NPs/TiO2 photoelectrode. However, the water splitting efficiency was very low because the monolayer of gold nanoparticles absorbed less than 50% of the incident light at a peak wavelength, and only about 10% of the visible light can be utilized, which can be estimated from the wavelength region of the absorption band. “In order to solve this light-harvesting problem, we focused on 'strong coupling' in the present study,” says Professor Hiroaki Misawa in the Research Institute for Electronic Science at Hokkaido University, who led the research. “Our photoelectrode consists of a sandwich structure by depositing a 30-nanometer titanium dioxide thin film on a 100-nanometer gold film and partially inlaying gold nanoparticles with an average size of 12 nanometers on the surface of the TiO2 thin film.”
The professor explains that because the TiO2/Au-film works as a Fabry-Pérot nanocavity, visible light is trapped by the cavity, given adequate thickness of the titanium dioxide. On the other hand, visible light is resonant with gold nanoparticles based on LSPR. “Importantly, in the present study, the nanocavity mode and plasmon mode are strongly coupled with each other when the two resonant wavelengths are close, which is named modal strong coupling,” Misawa noted. Therefore, hybrid modes between cavity and plasmon are formed. Because light scattering is small in Au-NPs used in this study (~12 nm) and TiO2 does not absorb visible light, up to 85% of visible light are finally harvested by the gold nanoparticles.
First demonstration of modal strong coupling applied to highly efficient plasmon-induced chemical reaction
Misawa’s team ventured that manipulating the plasmonic properties of noble metal nanoparticles in the strong coupling regime might be promising. “Based on this idea, we designed a Fabry-Pérot nanocavity of TiO2 thin-film/Au-film, and partially inlaid gold nanoparticles, realizing a modal strong coupling between the Fabry-Pérot nanocavity mode and LSPR.” He says few important works on the coupling between LSPR and the cavity had previously been reported, and only optical properties of the strong coupling systems were demonstrated. “To our knowledge, our present work is the first experiment that demonstrates that the modal strong coupling can be applied to the highly efficient plasmon-induced chemical reaction.”
Enhanced water-splitting process
The modal strong coupling, Misawa’s team considers, enhances the generation of plasmon-induced hot electrons. In typical plasmon-induced chemical reaction systems, the plasmon-induced hot electron generation efficiency is low. “In the present study, we employed a modal strong coupling system to successfully enhance the generation of plasmon-induced hot electrons,” he says.
Impact on the next generation of solar cells and other light technologies
The new photoelectrode developed by the Japan team could influence the design of the next generation of solar cells, Misawa agrees. He says to develop solid-state photovoltaics using their photoelectrode, the only engineering required would be depositing a hole transport layer and a transparent electrode (e.g., indium-tin-oxide) on the top of the gold-nanoparticle-spiked TiO2/Au-film.
Regarding his new photoelectrode’s impact on the design of other future light-based technologies Misawa says: “We expect that our achievement can provide a new design principle of light harvesting systems with a very small amount of materials and a strategy to accelerate the plasmon-induced chemical reactions under a strong coupling regime.”
Key applications
“For the energy harvesting using our photoelectrode, as demonstrated in the present work, the production of hydrogen fuel can be one of the promising applications of this technology,” Misawa projects. In addition, their contining work aims toward successful ammonia photosynthesis using atmospheric nitrogen and water, as well as photochemical carbon dioxide fixation.
Surprising outcome
Misawa admits he and his colleagues were “really surprised” by some of the results presenting during the research. “For example, we were surprised by the result that we obtained the enhancement of internal quantum efficiency (IQE) by 1.5-folds compared with the Au-NPs/TiO2 photoelectrode at a peak wavelength of IQE spectra under strong coupling conditions,” he says, elaborating that the IQE usually should not change, even if the absorption changes in a general photochemical reaction system. “We considered a quicker electron release under strong coupling conditions, which is deduced by the fact that the lifetime of surface plasmons is shorter than that of uncoupled Au-NPs, which leads to the enhancement of IQE.”
Challenges during the study
Misawa’s team faced two major challenges during the research study:
- One challenge is to design a strong coupling system that can be used for the plasmon-induced water splitting. In a typical strong coupling between cavity and matters, molecules or quantum dots are dispersed in the cavity. Actually, Au-NPs completely imbedded in the TiO2 thin-film/Au-film nanocavity cannot be used for the photochemical water splitting because Au-NP/TiO2/water interface is necessary for the water oxidation by the remaining holes, as described in the study. “Alternatively, we designed a partially inlaid Au-NP into the TiO2 thin-film/Au-film nanocavity to achieve the strong coupling between the Fabry-Pérot nanocavity-mode and LSPR while the plasmon-induced water splitting occurred on the three-phase boundary (Au-NP/TiO2/water). Based on this novel design, we successfully enhance the internal quantum efficiency of plasmon-induced photocurrent generation, employing water molecules as an electron source by 1.5 times under strong coupling conditions,” Misawa says.
- The other challenge was to understand the mechanism why the internal quantum efficiency of plasmon-induced water splitting is also enhanced. “We employed time-resolved photoelectron emission microscopy (PEEM) to characterize the lifetime of surface plasmons under strong coupling and uncoupled conditions,” the researcher says, reporting that under the strong coupling condition, a quicker electron release is expected, which is deduced by the fact that the lifetime of surface plasmon is shorter than that of uncoupled gold nanoparticles.
Next step
For the next step of their research, Misawa and his team want to explore the mechanism of strong coupling enhanced plasmon-induced charge formation and separation, especially the charge transfer dynamics and the water oxidation (oxygen evolution) mechanism. Furthermore, they also aim to expand the applications of their new photoelectrode to other light-energy conversions, such as ammonia photosynthesis, photochemical carbon reduction and solid-state solar cells.
The study, titled “Enhanced water splitting under modal strong coupling conditions,” was published in Nature Nanotechnology.
Written by Sandra Henderson, research editor Novus Light Technologies Today


















 Back to Features
Back to Features




























