Researchers from the universities of Bonn and Regensburg (Germany) have developed a novel type of organic light-emitting diode (OLED) suited to the design of particularly energy-efficient, inexpensive displays.
While OLEDs offer an especially bright image with high contrast, the problem with existing technologies, though, is that only about one quarter of the energy a device uses is actually converted into light. Manufacturers have been boosting this energy-to-light ratio by adding expensive, rare noble metals such as platinum or iridium to the active material. In a complete departure from current metal-organic OLED concepts, the team in Germany is now trying a different approach.
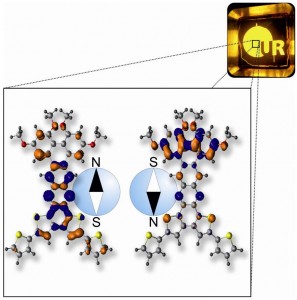
“In OLEDs, charges can recombine in two spin configurations, called singlets and triplets,” explains Professor John Lupton, Chair at the Institute of Experimental and Applied Physics, Faculty of Physics, at the University of Regensburg. Conventional OLEDs use either only singlets or triplets, not both. Lupton and his collaborators have now designed a novel molecule that can generate light both from singlets and triplets, simultaneously.
“The trick in our molecules is that we effectively freeze out any other relaxation pathway, so that triplets have to generate light, rather than heat,” says Lupton, who is also head of the Nanoscale Optoelectronics Group at the University of Utah (US). Instead of relying on adding expensive, rare metals, “We just bring patience,” the expert says. “In metal compounds, the lifetime is of the order of one microsecond. Our devices emit over 1000 times more slowly, but that does not matter, because — for reasons that are still not completely clear — they do not generate heat.”
Hence, the OLEDs developed by Lupton and his colleagues simultaneously generate light from singlets and triplets — or antiparallel and parallel spins. The professor adds that this is also important for retaining the spin information in OLEDs. “You can use electron spin as an information carrier. That is the principle of ‘organic spintronics’,” he says. “We show that you do not necessarily have to use expensive metals to get light out of triplets, there are other pure hydrocarbon tricks.”
For now, Lupton deems the impact of his team’s research success is mostly academic. “We are very excited that we have a molecule that emits both singlets and triplets. This could be useful in spintronics.” What surprises him the most, however, is “that the trick works and that nobody tried it before.” What is more, this was a chance discovery. His team was actually working on systematically tuning the energy gap between singlets and triplets when they realised this could apply to OLEDs, too. And it did. Plus, triplets are not always killed at room temperature, as previously assumed.
“One issue with our technology is that the triplet lifetime is very long — milliseconds. So you get a very fast build-up of triplet density,” Lupton says. “This will lead to non-linear interactions (i.e., triplet-triplet annihilation), which pose a loss channel. If the materials are used in applications, these will have to be operated at low current densities, such as in low-level background lighting.”
In conclusion, the researcher acknowledges their quantum efficiencies are extremely low, making it unlikely the technology will become viable anytime soon. “But on the other hand, this is a clear paradigm shift,” he says. “There is a wealth of early literature in molecular spectroscopy which we could turn back to and search for possible OLED material candidates.”
Research findings are detailed in the article “Metal-Free OLED Triplet Emitters by Side-Stepping Kasha’s Rule,” published in Angewandte Chemie, International Edition.
Image: Novel molecules for OLEDs. The synthetic design promotes random changes in orientation of the molecular "compass needle", increasing overall brightness.
Written by Sandra Henderson, Research Editor, Novus Light Technologies Today


















 Back to Features
Back to Features





























