Led by University of Rochester Medical Center researcher Benjamin Miller, the $1.7 million project is funded by the US Department of Defense Manufacturing Technology Program using CARES Act funds through a contract with AIM Photonics. The collaboration also involves Ortho Clinical Diagnostics, which develops and manufactures innovative laboratory testing and blood-typing solutions at its Global Center for R&D Excellence in Rochester; Syntec Optics, a maker of polymer optics in Rochester; researchers at the NY CREATES 300mm microelectronics research facility in Albany, New York, and at the University of California at Santa Barbara; and the Naval Research Laboratory in Washington, DC.
“This is a completely new diagnostic platform,” says Miller, the Dean’s Professor of Dermatology and a professor of biomedical engineering, optics, and biochemistry and biophysics. “We think this is going to be valuable in very broad applications for clinical diagnostics, not just COVID-19.”
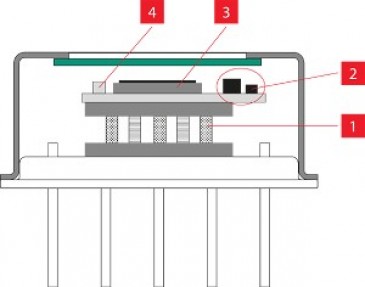
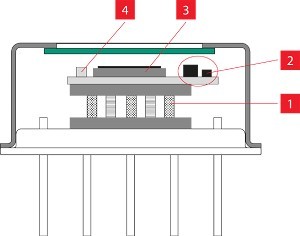
Key to the technology is an optical chip, no larger than a grain of rice. Proteins associated with eight different viruses, including SARS-CoV-2, are contained in separate sensor areas of the chip. If someone has been exposed to any of the viruses, antibodies to those viruses in the blood sample will be drawn to the proteins and detected.
Antibodies are proteins produced by the immune system to fight off specific bacteria and viruses. They remain in the immune system even after a patient recovers from an infection.
“It is exciting to see the sensors work developed by AIM Photonics, over the past five years, now play a part in more effective testing for COVID-19 and future diseases,” says Michael Cumbo, CEO of AIM Photonics. “The industry, academic, and government partnership is a fundamental piece of this institute. Together, we foster successful technology developments such as this optical chip, which in turn enables a very innovative diagnostic platform.”
The card will enable clinicians not only to detect and study COVID-19, but also to better understand potential relationships between COVID-19 infection and previous infections and immunity to other respiratory viruses, including circulating coronaviruses that cause the common cold.
“But one of the attractive aspects of this is there’s a pathway for this technology to eventually be used in a doctor’s office or a pharmacy,” Miller says.
“Our goal is to have a validated benchtop prototype by this winter, early spring at the latest.”
The researchers will use blood drawn from 100 consenting convalescent COVID-19 patients to test the device’s effectiveness.
When the researchers complete and validate the initial prototype, they will be able to apply for up to $5.3 million in additional funding to move the technology closer to commercial availability.



















 Back to News
Back to News