In the most powerful X-ray facilities in the world, scientists can analyze samples so small they contain only 10,000 atoms. Smaller sizes have proved exceedingly difficult to achieve, but a multi-institutional team has scaled down to a single atom.
“X-ray beams are used everywhere, including security scanning, medical imaging and basic research,” said Saw Wai Hla, physicist in the U.S. Department of Energy’s (DOE) Argonne National Laboratory and professor at Ohio University. ​“But since the discovery of X-rays in 1895, scientists have not been able to detect and analyze just one atom. It has been a dream of scientists to be able to do so for decades. Now we can.”
As just announced in Nature, scientists from Argonne and several universities report being able to characterize the elemental type and chemical properties of just one atom by using X-ray beams. This new capability will impact fundamental research in numerous scientific disciplines and development of new technologies.
The results from X-ray beams yield a sort of fingerprint for the type of elements in a material. For example, the NASA Curiosity rover gathered small samples of sand on the Martian surface, then determined with X-ray analysis that their content is similar to volcanic soil in Hawaii.
“Being able to study one atom at a time will revolutionize X-ray applications to an unprecedented level, from quantum information technology to environmental and medical research.” — Saw Wai Hla, Argonne physicist and professor at Ohio University
Using powerful X-ray machines called synchrotron light sources, scientists can analyze samples as small as a billionth of a billionth of a gram. Such samples contain about 10,000 atoms. Smaller sizes have proved exceedingly difficult to achieve, but in an astonishing leap, the team managed to scale down their observations to a single atom.
“The word transformative gets bandied about a lot, but this discovery I believe is truly a major breakthrough,” Hla said. ​“I was so excited I could barely sleep as I imagined possible uses in the development of batteries and microelectronic devices and even in environmental and medical research.”
To characterize just one atom with X-rays, it needs to be isolated from the same kind of atoms. To do so, the team first entwined a single iron atom in a nanometer-size molecule composed of different elements.
They then took the sample for analysis with the powerful X-ray beam at Argonne’s light source, the Advanced Photon Source (APS). The team detected the single atom in the sample at a beamline (XTIP) shared by the APS and the Center for Nanoscale Materials (CNM). Both are DOE Office of Science user facilities at Argonne. The beamline includes a scanning tunneling microscopy (STM) probe.
“A DOE Early Career Research Program Award that I received in 2012 allowed me to form a team of passionate scientists and engineers to develop the microscopy technique used in this study,” said Volker Rose, physicist at the APS and in the CNM. ​“Together, we developed and built this one-of-a-kind microscope at the XTIP beamline thanks to additional DOE funding.”
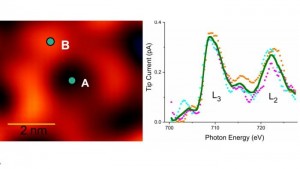
The rush of photons from the X-ray beams bombard the sample, causing it to release electrons. Positioned less than a nanometer above the sample surface, the STM probe collects the electric signal due to the emitted electrons. The resulting spectra (plots of current versus photon energy) are ​“fingerprints” for the elements in the periodic table. Each element has a unique fingerprint. By probing the sample surface, scientists can thus identify an element of a particular atom and its exact location.
There is more. They can also determine the atom’s chemical state from the same spectrum. The chemical state has to do with the fact that atoms can lose a certain number of electrons; for example, iron can lose two, three or four electrons. The chemical state reflects the number of electrons missing and is important for scientists to know because it affects the physical, chemical and electronic properties of the atom.
To prove the new capability’s wider applicability, the team successfully repeated the same X-ray analysis with terbium, a rare earth element. Rare earths are critical to microelectronics, batteries, aircraft structures and more. The technique is applicable to elements besides metals as well. By knowing the properties of single atoms, scientists can then exploit their uses in materials in new ways.
“Being able to study one atom at a time will revolutionize X-ray applications to an unprecedented level, from quantum information technology to environmental and medical research,” Hla said.
In addition to Hla and Rose, other Argonne authors to the Nature paper include Tolulope M. Ajayi, Nozomi Shirato, Tomas Rojas, Sarah Wieghold, Kyaw Zin Latt, Daniel J. Trainer, Naveen K. Dandu, Sineth Premarathna, Daniel Rosenmann, Yuzi Liu and Anh T. Ngo. Contributors from Ohio University include Xinyue Cheng, Sanjoy Sarkar, Shaoze Wang and Eric Masson. Other contributors are Xiaopeng Li, Shenzhen University; Yiming Li, University of South Florida; and Nathalie Kyritsakas, University of Strasbourg.
This research was funded by the DOE Office of Basic Energy Sciences. Computing resources were provided by the Laboratory Computing Resource Center at Argonne.
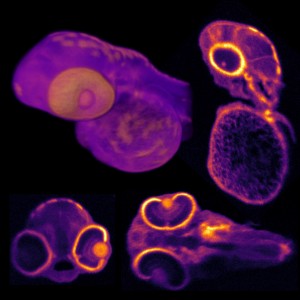
Image: Left: Image of a ring-shaped molecular host that contains just one iron atom. Right: X-ray absorption spectrum of single atom detected at location B in the molecular ring. Spectrum matches that of iron. Image by Argonne National Laboratory.



















 Back to News
Back to News



























