For years, the full utility of laser plastic welding for medical devices and other products was somewhat limited due to a simple design constraint: the requirement that laser-welded plastic assemblies comprise one “clear” or “transmissive” plastic part and a second “dark” or “absorptive” plastic part. This requirement ensured that, when laser energy was applied, the heat energy from the laser would pass through the transmissive part and accumulate in the weld zone of the absorptive part. There, the heat could soften the mating edges of both parts, enabling them to be brought together under compressive force into a clean, particle-free welded assembly.
But now, thanks to a new process, it is possible to laser weld two optically clear plastic parts made of a wide range of polymer materials for medical and other applications. This innovation combines Emerson’s Branson Simultaneous Through-Transmission Infrared (STTIr) laser-welding technology with precision ultrasonic spray-deposition technology from Sono-Tek. The breakthrough opens up new design possibilities for the makers of medical devices, drug delivery systems and in-vitro testing products.
The new “clear-on-clear” laser-welding process overcomes the traditional transmissive/absorptive part limitation by precision-treating one of two “clear” mating parts with a biocompatible laser absorber prior to laser welding. The absorber consists of microparticles of pigment dye or carbon black that are suspended in a carrier fluid such as isopropyl alcohol or acetone.
There are several methods for applying the laser absorber to clear plastic parts prior to welding. The absorber can be added to resin in the form of a masterbatch prior to molding, applied to molded parts using pad printing or, for maximum precision, applied to individual parts using Sono-Tek ultrasonic spray-deposition equipment. Spray deposition involves use of a precision ultrasonic atomizer to deposit laser-absorbing dye on one of the mating parts, creating a spray pattern width as small as 0.5 millimeter. When deposited using an optimal ultrasonic frequency and spray pattern, the carrier fluid essentially flashes off, leaving a precise pattern of laser-absorbing particles, one micron or smaller in size, on the surface of the thermoplastic. During the welding process, laser energy is absorbed by these particles and consumes them, releasing heat energy that conducts through the weld zone of the mating parts, which are then bonded together under compressive force.
According to Anita Ennest, regional sales manager for Sono-Tek, the ultrasonic spray-deposition systems “are designed to deposit minute quantities of pigment dye, measured in microliters, with coating thicknesses that are evaluated in the hundreds of nanometers on the surface of the coated parts.” She adds that the equipment used to apply the laser-absorbing suspension is modeled on earlier equipment developed to deposit ultra-thin, ultra-precise coatings on medical stents.
Ennest adds that the laser-absorbing suspension essential to the success of the new plastic welding process found its roots in light-absorbing coatings originally developed for use on photovoltaic cells, but is formulated with a biocompatible solvent and pigment dye combination. Developing the right combinations involved an iterative process that required the technology partners to transition the coating, deposition and weld processes from absorbent to transmissive materials, then transmissive to fully transparent materials.
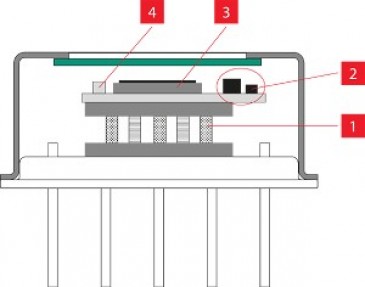
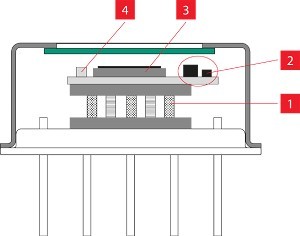
Another challenge facing the process was the need to dispense and deposit the laser-absorbing dye into precisely the right location on the joint of an injection molded part. Typical targets include the interior surface of a tongue-and-groove joint, or the interior surface of a “double V” joint on one of the mating parts (Figure 1). Precisely depositing the absorber within enclosed joints like these not only focuses the laser-generated heat needed to produce the part-to-part melt but also creates a “flash trap” that contains the treated area of the melt, isolating it from any nearby microfluidic flow paths in the welded part.
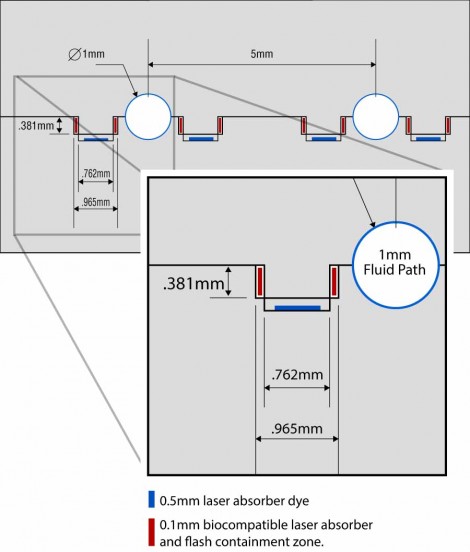
Figure 1: Tongue-and-groove laser joint example showing the biocompatible laser absorber and flash containment zone. Image courtesy of Emerson.
As Sono-Tek perfected the means of applying the laser absorber, Emerson maximized the effectiveness of the spray-enabled laser-welding process. The new clear-on-clear plastic laser-welding technology can reliably produce complex fluid paths while maintaining superior aspect ratios in the sub-millimeter flow path pitch. Such a degree of precision is essential for microfluidic parts, which must be reliably bonded yet maintain consistent dimensions along extremely small flow paths (Figure 2).

Figure 2: Two clear mating parts can now be laser welded using an innovative new welding process that can reliably produce complex fluid paths while maintaining superior aspect ratios. Images courtesy of Emerson.
Due to the precision of the spray-deposition process, there’s a reduced need to precisely image the laser energy, since clear-on-clear welding only occurs where the laser-absorbing dye has been deposited. Additionally, when a tongue-and-grove joint or a similar joint design is employed within the mating parts, the joint can be used to properly align the entire assembly prior to welding. And, as noted, the area containing the finished weld can be completely isolated from the functional microfluidic path in the completed assembly — a must for medical or medical device applications.
New technology combines with many existing laser-weld processes
Because this new “clear-on-clear” laser-welding capability relies on equipment that is added to the STTIr laser-welding process — the biocompatible laser absorber and ultrasonic spray-deposition equipment — it can be “bolted on” to many existing laser-welding applications. Then, once implemented, it delivers cycle times measured in seconds, making industrial-scale laser welding of medical-quality clear-on-clear microfluidic parts economically viable for the first time. And, since the spray-deposition process can be adapted to process multiple parts at once, both the spray-deposition and welding processes can be further scaled up to accelerate production throughputs.
The new process also allows for a far greater range of clear thermoplastics to be combined, regardless of chemistry. Because the laser-absorbing pigment dye so efficiently generates and conducts heat and melt, its use can compensate for even larger differences in the glass transition temperature (Tg) of different polymers used in mating parts.
A typical example of the process involves the use of two clear plastic “coupons” — injection-molded parts that, together, mate to form a serpentine flow path like those found in in-vitro diagnostic (IVD) systems, human drug-delivery systems or implantable medical devices. The coupons that will become the “absorptive” half of each assembly are placed into the Sono-Tek spray-deposition equipment to deposit the laser absorber.

Figure 3: The Branson GLX Micro laser welder from Emerson is the smallest of five units in the GLX Series. The welding unit (left) features a color touchscreen HMI, a lift table measuring 150 x 150 mm, and clamp forces up to 0.05kN, making it an excellent choice for welding microfluidic parts used in medical applications. The unit at right contains the welder’s laser source and power supply. Image courtesy of Emerson.
From there, the parts are transferred to a laser welder (Figure 3), where the result is a clear-on-clear, laser-welded part. For medical or IVD devices that demand optically clear flow paths, the technology is invaluable, simplifying everything from automating blood cell counts in capillary-sized fluid paths to providing visual validation to a technician that a microdose of a powerful therapy is being properly administered to a patient.

Figure 4:By applying a solvent-based laser absorber to the mating surface of one part, Emerson’s STTIr laser-welding technology can now join clear-on-clear parts with exceptional aesthetic and functional results. Image courtesy of Emerson.
Written by Tom Hoover, Sr. Medical Business Development Manager, Americas, for Branson at Emerson



















 Back to Features
Back to Features