The superior precision and flexibility of laser processing have resulted in its ever widening use in the fabrication of medical devices - implants and surgical tools, from simple hole drilling in catheters to sophisticated three-dimensional cutting of stents. Now, lasers are poised to enable certain types of cardiac intervention to advance from open chest surgeries to minimally invasive procedures. Specifically, three different types of replacement heart valves are at various stages of regulatory approval in Europe and the U.S. These are hybrid valves based on a metal scaffold using nitinol (often called memory metal) which is cut with a fiber laser-based system.
The need for heart valve replacement
The heart contains four valves whose operation is critical for circulating blood to the lungs and around the human body. Some patients have congenitally defective heart valves. And many others suffer from valve disease due to a variety of problems such as chronically elevated blood pressure and chronic obesity. Even prior steroid use in body builders has been implicated as a culprit leading to valve deterioration and damage.
In recent decades, the medical research community has explored numerous valve replacement options to treat these patients; to restore quality of life and/or prevent terminal decline. Denatured animal (usually porcine) valves have been used to replace defective mitral valves. Other replacements have included mechanical aortic valves and more recently, engineered tissue valves created from the patient’s own stem cells, using some type of synthetic or animal sourced scaffold material. Each of these has some type of limitation. For instance, mechanical valves can cause clotting and require the patient to take anti-coagulant drugs, whereas the tissue engineered valve tends to shrink over time and then leak as the tissue is slowly resorbed and naturally replaced. In addition to these functional limitations, there is also the challenge of surgical insertion; most heart valve replacement requires open heart surgery. This is expensive, risky and painful. Moreover, many patients are not suitable candidates for open heart surgery because of the state of their heart disease, advanced age or other health factors. Several new prosthetic heart valves have been developed to address these limitations, and more importantly, to allow transcatheter insertion.
Laser cutting of nitinol scaffolds
These valves are multilayered devices based on a nitinol scaffold. STI Laser Industries (Or Akiva, Israel) is a medical device manufacturer specializing in laser cutting, micromaching and finishing of medical devices. They are a key manufacturer of these nitinol heart valve frames. Tovy Sivan, CEO & President of STI explains, “STI has a long history of fabricating medical devices with an emphasis on both laser cutting and welding. We use several different laser types because of the diversity of devices and materials. These range from the original flashlamp-pumped Nd:YAG lasers with which we began cutting metal stents 25 years ago, to today’s ultrafast lasers that we use to micromachine plastic disposables. Virtually all of these lasers have been supplied by Rofin (now Coherent).” Sivan believes that these new hybrid heart valves are poised to dramatically change heart valve replacement surgery with improved outcomes, lower costs and high patient satisfaction.
The use of a nitinol frame is key to the ability to insert these valves in a minimally invasive transcatheter procedure. Nitinol, an alloy of nickel and titanium is a type of “memory metal.” After machining, a structure made from a memory metal is thermally treated in order to permanently fix or set the shape. If the metal structure is then twisted, bent, or otherwise physically deformed, it will later return to its preset shape when it is warmed above its phase transition temperature – see figure 1. By optimizing the nickel/titanium ratio and the use of minor metal additives, this transition temperature can be chosen to be only 10-20 ºC below body temperature.

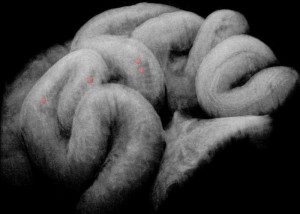
Heart valve scaffolds are laser cut from a nitinol tube blank. Image courtesy of STI Laser Industries.
Sivan explains, “Traditional replacement heart valves are simply too large to insert through a catheter. But with a nitinol frame, the valve can be mechanically collapsed so that its diameter is reduced by a factor of five. This means even a large valve, like the mitral valve, can be introduced through a catheter inserted through the femoral artery in the groin. Once it has been placed into the heart through its apex, the valve can then be gently extracted out of the delivery system, and once exposed to body temperature it springs back into its set shape ready for anchoring in place.” He notes that the nitinol metal is only the frame; STI supplies these custom nitinol structures to a medical device manufacturer who then sutures the valve's leaflets and a cover of denatured animal (e.g., porcine) tissue. (The use of bare metal would increase the risk of blood clotting and valve stenosis).
Fiber laser affords accuracy, quality and more
STI already manufactures three types of heart implants from nitinol: aortic valves, mitral valves and shunts that relieve the pressure between the right and left atrium. These range in diameter from 10 mm to 30 mm. The frames for these valves are first cut at STI from a nitinol tube blank using the Coherent-Rofin StarCut Tube SL tube cutting machine equipped with a StarFiber 320 FC fiber laser (output at ~1 µm). This tool combines a static beam with tube motion to provide high accuracy three dimensional cutting. After cutting, STI shape-sets the frames to the desired scaffold configuration after which they are electro-polished to prepare them for application of the bio-compatible covering. Sivan summarizes the main advantages of fiber laser cutting, “This application needs accuracy, repeatability, and high surface quality. We also need to minimize process time for this highly complex shape cutting. Our fiber laser system meets all these needs and more.”
With regulatory approval already achieved for some of these valves in Europe and advanced trials approved by the FDA in the U.S., Sivan predicts this product will significantly improve outcomes for many patients with heart valve conditions.
Written by by Roland Wölzlein and Frank Gaebler, Coherent Inc.

























 Back to Features
Back to Features