Twenty years ago the only sure way to know whether a person had Alzheimer’s disease was by examining the brain through autopsy, after death. Thanks to advances in research over the past few decades, lab and imaging tests are now available to help a doctor or researcher see biological signs of the disease, or biomarkers, in a living person. Blood tests can measure levels of beta-amyloid, a protein that accumulates abnormally in the brains of people with Alzheimer’s. And recent advances in imaging techniques, however, are offering a variety of options.
According to the Mayo Clinic, commonly used imaging techniques include:
- Magnetic resonance imaging (MRI). An MRI uses powerful radio waves and magnets to create a detailed view of your brain.
- Computerized tomography (CT). A CT scan uses X-rays to obtain cross-sectional images of your brain.
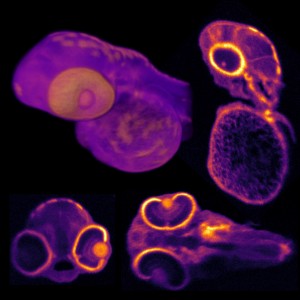
- Positron emission tomography (PET). A PET scan uses a radioactive substance known as a tracer to detect substances in the body. There are different types of PET scans. The most commonly used PET scan is a fluorodeoxyglucose (FDG) PET scan. This scan can identify brain regions with decreased glucose metabolism. The pattern of metabolism change can distinguish between different types of degenerative brain disease.
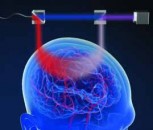
While imaging may offer information about plaques in the brain, conducting imaging tests on patients with signs of cognitive impairment is often a difficult task and can be frightening and uncomfortable for the patient. However, a team of researchers in Europe have found a way of replacing two separate imaging scans with one, which cuts the time of the procedure—making patients more comfortable—but it also reduces cost and radiation exposure.
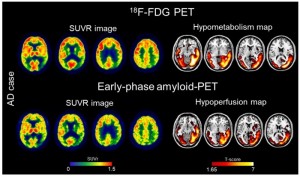
The novel nuclear medicine imaging protocol is called dual-phase amyloid PET, and the advantage over traditional scans is that with a single tracer injection, physicians can assess both amyloid deposition and neurodegeneration.
The currently used F-FDG (fluorodeoxyglucose) scans are used to measure brain metabolism and are performed to evaluate beta-amyloid deposition, which is an indicator of Alzheimer’s. They require much patience because the patient must be still for up to two hours after the radioactive tracer is injected.
“During the amyloid PET scans, nuclear medicine physicians wait at least 50 minutes after the radiotracer injection to acquire the image of beta-amyloid deposition. If images are also taken immediately after injection (early-phase), they can provide a measure of brain perfusion. Since brain perfusion and brain metabolism are closely related, the perfusion information could replace an 18F-FDG PET scan,” said Cecilia Boccalini, MSc, PhD candidate at the Laboratory of Neuroimaging and Innovative Molecular Tracers and faculty of medicine at the University of Geneva Neurocenter in Geneva, Switzerland and faculty of neuroscience at Vita-Salute San Raffaele University in Milan, Italy.
The researchers used both methods to evaluate 166 patients who had a range of cognitive impairment. The team created brain hypometabolism maps of each patient in order to compare early- patients along the Alzheimer’s phase amyloid PET scans and 1F-FDG scans.
What they found was that brain hypoperfusion assessed by early-phase amyloid PET is comparable with brain hypometabolism assessed by F-FDG-PET. The findings show cost-saving capacities of amyloid PET and the researchers concluded that it "supports routine use of the modality for individual classification in clinical practice".
“This dual-phase amyloid-PET protocol allows physicians to measure brain amyloid and perfusion in a single procedure,” stated Valentina Garibotto, MD, head of the Nuclear Medicine and Molecular Imaging Division at Geneva University Hospitals in Geneva, Switzerland, and associate professor at the University of Geneva. “Obtaining both amyloid and perfusion data with a single tracer injection is optimal in terms of patient safety and radiation exposure, comfort, and costs. Our work supports the routine use of the dual-phase amyloid PET protocol in clinical practice.”




















 Back to Features
Back to Features

























