Researchers at the Research Institute of Molecular Pathology (IMP) and the Max F. Perutz Laboratories (MFPL) in Vienna, Austria, collaborated with scientists at the Massachusetts Institute of Technology (MIT) in Massachusetts, US, to create an imaging system that reveals neural activity in living animals.
This may be the first technique that can generate 3D movies of entire brains at the millisecond timescale, and it could help to discover how neuronal networks process sensory information and generate behavior. The new approach is described in Simultaneous whole-animal 3D-imaging of neuronal activity using light-field microscopy., published online by Nature Methods on 18 May, 2014.
Worms and fishes
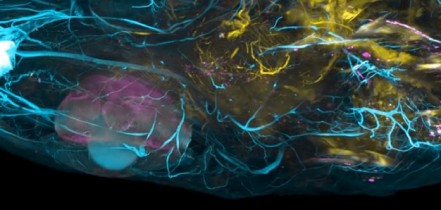
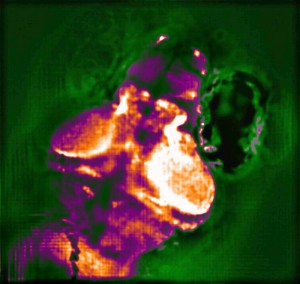
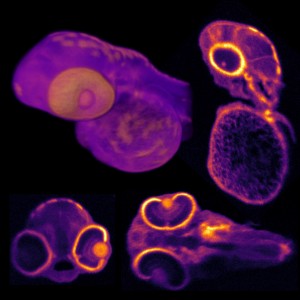
The team used the new system to simultaneously image the activity of every neuron in the worm Caenorhabditis elegans, as well as the entire brain of a zebrafish larva, offering a more complete picture of nervous system activity than has been previously possible. The used the worm because it is the only organism for which the entire neural wiring diagram is known. This one-millimeter worm has 302 neurons, each of which the researchers imaged as the worm performed natural behaviors, such as crawling.
They studied larvae of zebrafish to demonstrate the power of the new technology in higher organisms. Their nervous system consists of over 100 000 neurons that fire at a much faster rate, rather like humans. In the tiny larvae, the scientists were able to induce neuronal response to odor stimuli in around 500 neurons and track the nerve signals simultaneously in about 5000 activated neurons.
Vaziri’s team developed the brain-mapping method together with researchers in the lab of Edward Boyden, an associate professor of biological engineering and brain and cognitive sciences at the Massachusetts Institute of Technology (MIT).
Neurons encode information including sensory data, motor plans, emotional states, and thoughts, using electrical impulses called action potentials, which provoke calcium ions to stream into each cell as it fires. By engineering model organisms that carry fluorescent proteins that glow when they bind calcium, scientists can visualize this electrical firing of neurons in live animals. However, until now there has been no way to image this neural activity over a large volume, in three dimensions, and at high speed.
Scanning the brain with a laser beam can produce 3D images of neural activity, but it takes a long time to capture an image because each point must be scanned individually. The research-team wanted to achieve similar 3D functional images but accelerate the process so they could see neuronal firing, which takes only milliseconds, as it occurs.
The new method is based on a technology known as light-field imaging, which creates 3D images by capturing angular information of incoming rays of light. In the new paper, the researchers in Vienna and Cambridge built a light-field microscope that was optimized to have single neuron resolution and applied it, for the first time, to imaging of neural activity.
With this kind of microscope, the light emitted by the sample is sent through an array of lenses that refracts the light in different directions. Each point of the sample generates about 400 different points of light, which can then be recombined using a computer algorithm to recreate 3D structures.
Robert Prevedel, a postdoc in the Vaziri Lab and first author of the paper, built the microscope at the IMP in Vienna. Young-Gyu Yoon, a graduate student at MIT and co-first author, devised the computational strategies that reconstruct the 3D images.
The findings could be ultimately useful in developing new types of algorithms that simulate functions of the brain and predict behaviour. Such models are in high demand in the area of machine learning and object recognition and classification.


































 Back to News
Back to News